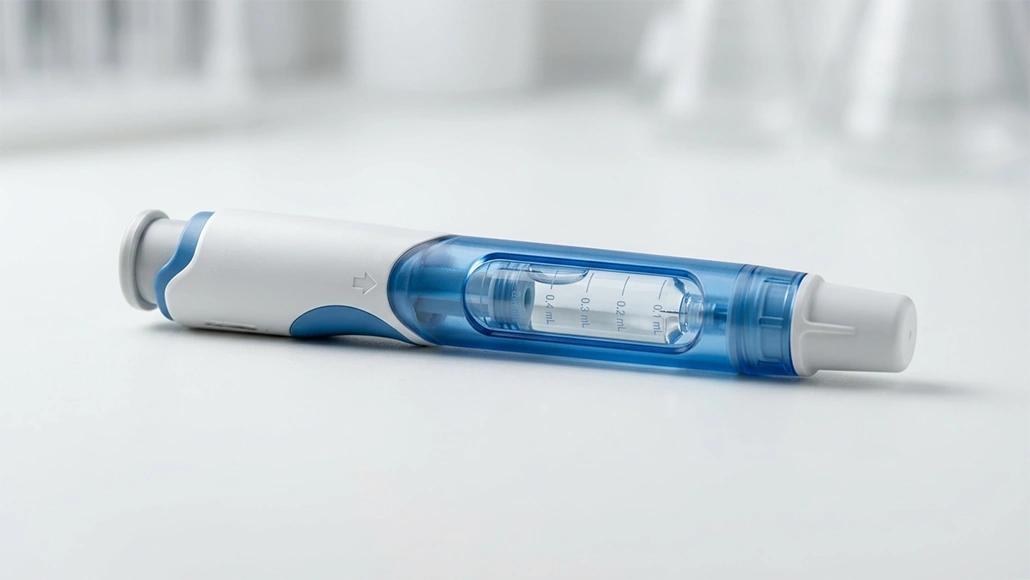
The pharmaceutical industry has undergone a radical transformation over the past two decades, shifting its focus from small-molecule drugs to complex biological products known as biologics. These therapies, which include monoclonal antibodies, vaccines, and gene therapies, are derived from living organisms and offer a level of specificity and efficacy that traditional chemical drugs simply cannot match. However, the very complexity that makes biologics so effective also makes them incredibly difficult to develop and stabilize. Biologics formulation challenges are numerous, ranging from the physical fragility of the molecules to the logistical difficulties of delivering them to the patient in a stable and active form. Unlike small-molecule drugs, which are typically robust and can be easily synthesized in a lab, biologics are large, three-dimensional structures that are highly sensitive to their environment. Even slight changes in temperature, pH, or mechanical stress can cause a protein to denature, lose its shape, and become ineffective or even dangerous. This inherent instability requires a highly sophisticated approach to protein formulation, where the goal is to create a protective environment that preserves the molecule’s structural integrity from the moment of production until it is administered to the patient. As the demand for these therapies grows, finding innovative solutions to these challenges has become a primary driver of pharma innovation.
Addressing Biologics Stability in High-Concentration Formulations
One of the most pressing biologics formulation challenges is the need for high-concentration formulations. Many biologics, particularly monoclonal antibodies for chronic diseases like rheumatoid arthritis, require high doses to be effective. Traditionally, these were administered through intravenous (IV) infusions in a hospital setting, which is both time-consuming and expensive. To improve the patient experience, there is a strong push toward subcutaneous (SC) delivery, which would allow patients to self-administer their medication at home. However, delivering a large dose in the small volume required for an SC injection means the drug must be highly concentrated. High-concentration protein formulation leads to a host of new problems, most notably a dramatic increase in viscosity. When proteins are packed tightly together, they tend to interact and stick to each other, creating a solution that is thick and difficult to pull through a needle. Furthermore, these high concentrations often lead to protein aggregation a process where proteins clump together. Aggregation is one of the most significant risks in biopharmaceutical development, as it not only reduces the drug’s efficacy but can also trigger a dangerous immune response in the patient. Innovative solutions, such as the use of specific amino acids and surfactants, are being developed to disrupt these interactions and keep the proteins in a stable, monomeric state even at high concentrations. This work is essential for making biologics more practical and less burdensome for patients managing long-term conditions.
Innovations in Drug Delivery Biologics and Device Integration
The delivery of biologics is as much a mechanical challenge as it is a chemical one. Because these molecules are easily broken down by the digestive system, they cannot be taken as pills and must almost always be injected. To overcome the limitations of traditional needles, drug delivery biologics is moving toward integrated systems like auto-injectors and wearable patch pumps. These devices are designed to handle the high viscosity of modern biologics, using motorized or spring-loaded mechanisms to ensure a smooth and consistent delivery. The integration of the drug and the device known as a combination product is a key trend in biopharmaceutical development. Furthermore, researchers are exploring “bio-inspired” delivery methods that could eventually allow for the oral delivery of biologics. This involves encapsulating the protein in a protective shell that can survive the harsh environment of the stomach and then use microscopic “microneedles” to inject the drug directly into the wall of the small intestine. While still in the early stages, these innovations represent a potential paradigm shift that would eliminate the need for injections entirely. By focusing on the interface between the biological molecule and the delivery device, pharma innovation is making these complex therapies more accessible and user-friendly for patients everywhere. We are also seeing the development of devices that can sense when the injection is complete and provide feedback to the patient, ensuring that the full dose is delivered every time.
Scalable Manufacturing and the Quest for Biopharmaceutical Robustness
The transition from a laboratory setting to large-scale production introduces another layer of biologics formulation challenges. The processes of filtration, pumping, and filling can all exert mechanical stress on delicate proteins, leading to degradation. Ensuring biologics stability during scalable manufacturing requires a deep understanding of fluid dynamics and thermodynamics. Companies are increasingly turning to “Quality by Design” (QbD) principles, where stability is built into the manufacturing process from the very beginning. This involves using advanced sensors and real-time monitoring to detect any signs of protein stress before it leads to a loss of quality. Lyophilization, or freeze-drying, remains a vital tool for ensuring the long-term stability of biologics. By removing water and locking the protein in a solid, “glassy” state, lyophilization prevents most chemical and physical degradation. However, the freeze-drying process itself can be stressful for the protein, requiring the use of specialized cryoprotectants like sucrose or trehalose. As we move toward more globalized supply chains, the development of stable, room-temperature formulations is a high priority. This would reduce the reliance on the “cold chain” and make these life-saving biopharmaceuticals easier to distribute in developing countries, where refrigeration may be inconsistent. This global perspective is essential for ensuring that the benefits of biotechnology are shared by all, regardless of where they live.
The Future of Biopharma and Personalized Stabilization
As the field of biopharma continues to evolve, we are seeing a move toward more personalized formulation strategies. Just as the drugs themselves are being tailored to specific patient populations, the way they are stabilized and delivered is also becoming more customized. Advances in computational modeling allow researchers to predict how a specific protein will behave in different environments, allowing for the rapid design of optimized formulations. This “digital twin” approach to biopharmaceutical development is significantly shortening the time it takes to bring new therapies to market while ensuring a higher level of safety and efficacy. The ultimate goal of overcoming biologics formulation challenges is to unlock the full potential of biological medicine. Whether it is a gene therapy that cures a rare genetic disorder or a monoclonal antibody that manages a chronic disease, these therapies represent the pinnacle of human ingenuity. By continuing to innovate in the areas of stability, delivery, and manufacturing, we are ensuring that the promise of biopharma is fulfilled for all patients. The journey from a living cell to a finished medication is long and complex, but with every challenge overcome, we move closer to a future where even the most complex diseases can be managed with precision and grace. This future is one where the complexity of the therapy is matched by the sophistication of its delivery, creating a seamless path from discovery to cure.
Collaborative Ecosystems for Biopharma Success
The complexity of biologics means that no single company can solve every formulation challenge in isolation. We are seeing a rise in collaborative ecosystems where biotech firms, device manufacturers, and academic researchers work together to develop integrated solutions. These partnerships are essential for bridging the gap between molecular biology and mechanical engineering. For instance, a biotech company might develop a promising new protein, but it requires a specialized delivery device to be commercially viable. By working together from the early stages of development, these partners can ensure that the final product is optimized for both stability and user-friendliness. Furthermore, these ecosystems foster the sharing of best practices and data, accelerating the overall pace of innovation in the industry. This collaborative approach is what will ultimately allow us to tackle the most difficult biologics formulation challenges and bring transformative new therapies to patients more quickly. It is a model for how the industry can work together to solve the most pressing health challenges of our time.
Navigating the Intellectual Property and Regulatory Landscape
As we develop new and innovative solutions for biologics, navigating the intellectual property (IP) and regulatory landscape becomes increasingly complex. Protecting the proprietary formulations and delivery devices is essential for ensuring that companies can recover their investments in research and development. However, we must also ensure that the patent system encourages innovation rather than stifling it. Regulators are also faced with the challenge of evaluating these complex combination products, requiring new frameworks that account for both the drug and the device. This dialogue between science, law, and policy is critical for creating an environment where biopharmaceutical innovation can thrive. By establishing clear and predictable pathways for approval, we can reduce the time and cost of bringing new biologics to market, ultimately benefiting patients through faster access to better treatments. This focus on the “business” of biopharma is a necessary complement to the scientific breakthroughs we’ve discussed, ensuring that the science translates into real-world impact.