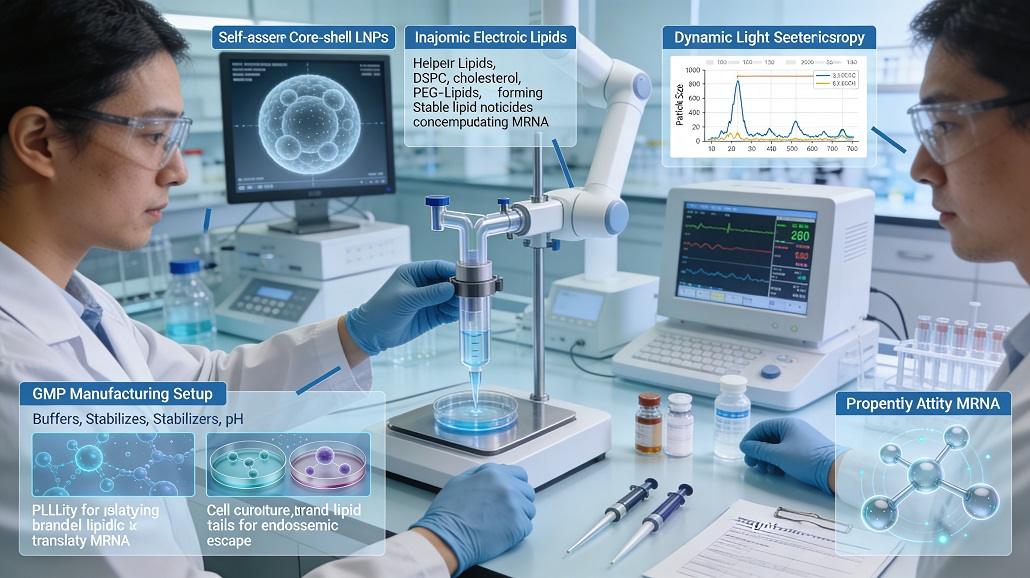
The landscape of modern pharmacology has been irrevocably altered by the emergence of messenger RNA as a potent therapeutic modality. While the potential of mRNA to instruct cells to produce proteins for vaccination or disease treatment is vast, its practical application depends entirely on the vehicle that carries it. Without a robust delivery system, mRNA is rapidly degraded by extracellular RNases and fails to cross the negatively charged cellular membrane. This challenge has placed lipid nanoparticles at the forefront of pharmaceutical research, where LNP formulation strategies for mRNA drug delivery have become the decisive factor in clinical success. These strategies involve a complex interplay of chemical engineering and biological insight, aiming to create a stable, non-toxic, and highly efficient transport mechanism.
At the heart of any effective lipid nanoparticle lies the ionizable lipid, a component that remains neutral at physiological pH but becomes positively charged within the acidic environment of the endosome. This pH-dependent switch is the primary driver of encapsulation efficiency and endosomal escape, two metrics that define the potency of an mRNA therapeutic. When developing LNP formulation strategies for mRNA drug delivery, the selection of the ionizable lipid dictates the entire pharmacokinetic profile of the drug. Modern strategies now prioritize lipids with branched tails and specific pKa values that minimize immunogenicity while maximizing the release of mRNA into the cytoplasm. This delicate balance is achieved through rigorous screening and the use of molecular modeling to predict how lipid headgroups interact with both the RNA cargo and the target cell membranes.
Structural Components and the Role of Helper Lipids
Beyond the ionizable lipid, the structural integrity of the nanoparticle is maintained by a precise mixture of helper lipids, cholesterol, and PEG-lipids. Each of these components plays a distinct role in the overall performance of the delivery system. Cholesterol, for instance, is essential for providing structural rigidity and filling gaps within the lipid bilayer, which directly impacts the stability of the nanoparticle during storage and circulation. Helper lipids, often phospholipids like DSPC, facilitate the transition of the nanoparticle into a stable lamellar or hexagonal phase, which is critical for the fusion of the LNP with the host cell membrane. The inclusion of these elements is a key part of LNP formulation strategies for mRNA drug delivery, ensuring that the particles remain intact until they reach their intended destination.
The final component, PEG-lipids, acts as a surface shield that prevents the aggregation of nanoparticles and inhibits their recognition by the reticuloendothelial system. By forming a “stealth” layer, PEGylated lipids extend the half-life of the drug in the bloodstream, allowing for more consistent dosing and improved patient outcomes. However, the concentration and chain length of the PEG must be carefully calibrated. Too much PEG can inhibit the cellular uptake of the LNP, a phenomenon often referred to as the “PEG dilemma.” Therefore, optimizing the molar ratio of these four primary components is a central pillar of LNP formulation strategies for mRNA drug delivery, requiring iterative testing to find the “sweet spot” where stability meets bioactivity.
Enhancing Encapsulation Efficiency and RNA Stability
One of the most significant hurdles in LNP development is ensuring that the mRNA cargo remains functional throughout the manufacturing process and subsequent delivery. High encapsulation efficiency is not merely about using less raw material; it is about ensuring that every nanoparticle carries a therapeutic payload that is protected from the external environment. Advanced LNP formulation strategies for mRNA drug delivery utilize precise mixing techniques to ensure that the electrostatic interaction between the negatively charged RNA and the positively charged ionizable lipids is maximized during the self-assembly process. This results in a dense, core-shell structure where the RNA is sequestered in the center, shielded from enzymatic degradation.
Furthermore, maintaining RNA stability requires an understanding of the chemical interactions within the nanoparticle core. Factors such as internal pH, moisture content, and the presence of impurities can all lead to the hydrolysis or oxidation of the mRNA. Modern formulation strategies incorporate specialized buffers and stabilizers that maintain a protective microenvironment within the LNP. This is particularly important for long-term storage and global distribution, where cold-chain requirements can be a significant logistical burden. By enhancing the intrinsic stability of the formulation, developers can extend the shelf life of mRNA products and reduce the reliance on ultra-low-temperature storage, making these life-saving therapies more accessible to diverse populations.
Overcoming the Challenges of Endosomal Escape
The effectiveness of any RNA-based therapy is ultimately determined by its ability to reach the cytoplasm. Once an LNP enters a cell via endocytosis, it is trapped within an endosome. If the nanoparticle cannot escape, the mRNA will be degraded in the lysosome before it can be translated into protein. Advanced LNP formulation strategies for mRNA drug delivery focus on optimizing the ionizable lipid’s ability to trigger endosomal membrane disruption. This is achieved by designing lipids that undergo a phase transition as the endosome acidifies, leading to the fusion of the LNP and endosomal membranes.
Researchers are exploring various lipid geometries and tail architectures to enhance this escape mechanism. Branched lipid tails, for instance, can create more “void space” within the lipid bilayer, facilitating the membrane fusion required for release. Additionally, the incorporation of specific helper lipids that promote the formation of non-lamellar phases has shown promise in improving the delivery efficiency of these systems. By focusing on the molecular mechanics of endosomal escape, scientists are able to lower the required dose of mRNA, thereby reducing the potential for systemic toxicity and improving the overall safety profile of the therapeutic.
Transitioning to GMP Manufacturing and Regulatory Readiness
As a formulation moves from the laboratory bench to the clinic, the focus shifts toward scalability and regulatory compliance. LNP formulation strategies for mRNA drug delivery must be designed with Good Manufacturing Practice in mind from the outset. This means selecting high-purity raw materials and developing processes that are reproducible at a large scale. The transition from small-batch production to commercial manufacturing often involves shifting to continuous flow processes, such as microfluidic mixing or T-junction mixing. These methods allow for precise control over the physical properties of the LNPs, such as particle size and polydispersity index, which are critical quality attributes monitored by regulatory agencies.
Regulatory readiness also involves comprehensive characterization of the LNP-mRNA complex. Health authorities require detailed data on the physical and chemical stability of the drug product, as well as its safety profile in pre-clinical models. By employing robust LNP formulation strategies for mRNA drug delivery, companies can provide the necessary evidence of consistency and potency required for clinical trial authorization. This includes demonstrating that the manufacturing process does not introduce harmful byproducts and that the final product maintains its efficacy across different batches. As the field matures, these strategies will continue to evolve, incorporating new lipid chemistries and innovative delivery routes to expand the therapeutic potential of mRNA beyond vaccines and into the realm of chronic disease management.
The Role of Analytical Development in Formulation Success
Success in LNP development is inextricably linked to the quality of the analytical methods used to measure performance. Advanced LNP formulation strategies for mRNA drug delivery rely on a suite of characterization tools that provide real-time feedback during the development process. Techniques such as dynamic light scattering and nanoparticle tracking analysis are used to monitor particle size and homogeneity, while high-performance liquid chromatography ensures the purity of the lipid components. Additionally, the use of cryogenic electron microscopy has become a standard for visualizing the internal structure of the LNPs, allowing researchers to confirm the presence of a stable, drug-loaded core.
Furthermore, potency assays that measure the translation of mRNA in cell-based models are essential for verifying the biological activity of the formulation. These assays provide a direct link between the physical properties of the nanoparticle and its therapeutic effect. By integrating these analytical tools into the formulation workflow, developers can rapidly identify the most promising candidates and refine their LNP formulation strategies for mRNA drug delivery. This data-driven approach not only accelerates the development timeline but also ensures that the final product is capable of meeting the rigorous demands of the clinical environment.
Future Perspectives in LNP Design and Delivery
Looking ahead, the next generation of LNP formulation strategies for mRNA drug delivery will likely focus on tissue-specific targeting and enhanced shelf-life stability. While current formulations are primarily effective at delivering mRNA to the liver, there is a significant need for delivery systems that can reach other organs, such as the lungs, heart, or brain. This will require the development of novel lipids and surface modifications that can bypass biological barriers and navigate the complex environment of the human body. Additionally, the development of lyophilized or heat-stable LNP formulations will be critical for expanding the reach of mRNA therapies to regions with limited cold-chain infrastructure.
As we continue to unravel the complexities of lipid-nanoparticle interactions, the potential for mRNA technology remains vast. By refining LNP formulation strategies for mRNA drug delivery, the scientific community is laying the groundwork for a new era of personalized medicine. From rare genetic disorders to common metabolic diseases, the ability to deliver therapeutic instructions directly to the body’s cells offers a revolutionary path forward for global healthcare. The journey from the lab to the patient is long and challenging, but with the right formulation strategies in place, the promise of mRNA can finally be realized on a global scale.